February 2023
Features
FDA 510(k) Clearance
MD.ai Viewer is now FDA-cleared for diagnostic use as a Class II medical device intended for display of DICOM and non-DICOM medical images and other healthcare data to aid in the diagnosis for healthcare professionals. More information can be found here.
Notes and Report Panel
Navigation panels can now be toggled to remain in place or continue as viewport overlays. By default the panel will be docked if sufficient screen space is available.
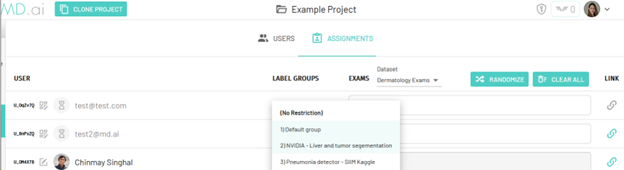
Add Users to Multiple Label Groups
In addition to assigning project users specific exams, project administrators can now restrict users to single or multiple label groups for blinding or strict annotation control. More detailed information can be found here.
Enhanced Cloned Project Functionality
Admins who allow their projects to be cloned can now also choose to allow users to upload data to cloned projects. This setting can be turned on by clicking the Allow users to update data in cloned project button in the project settings menu. The default remains that users of cloned projects cannot upload additional data to cloned projects.
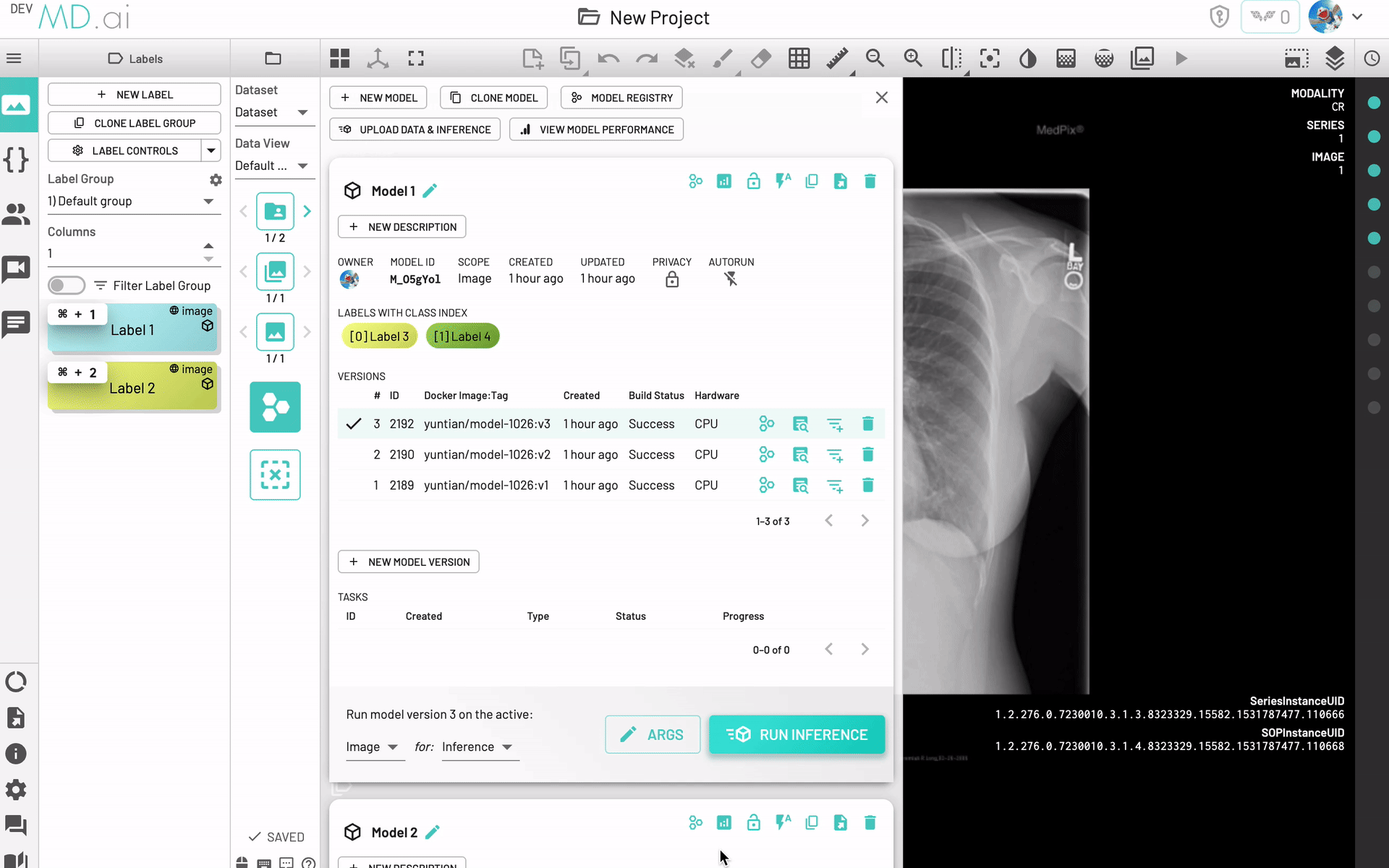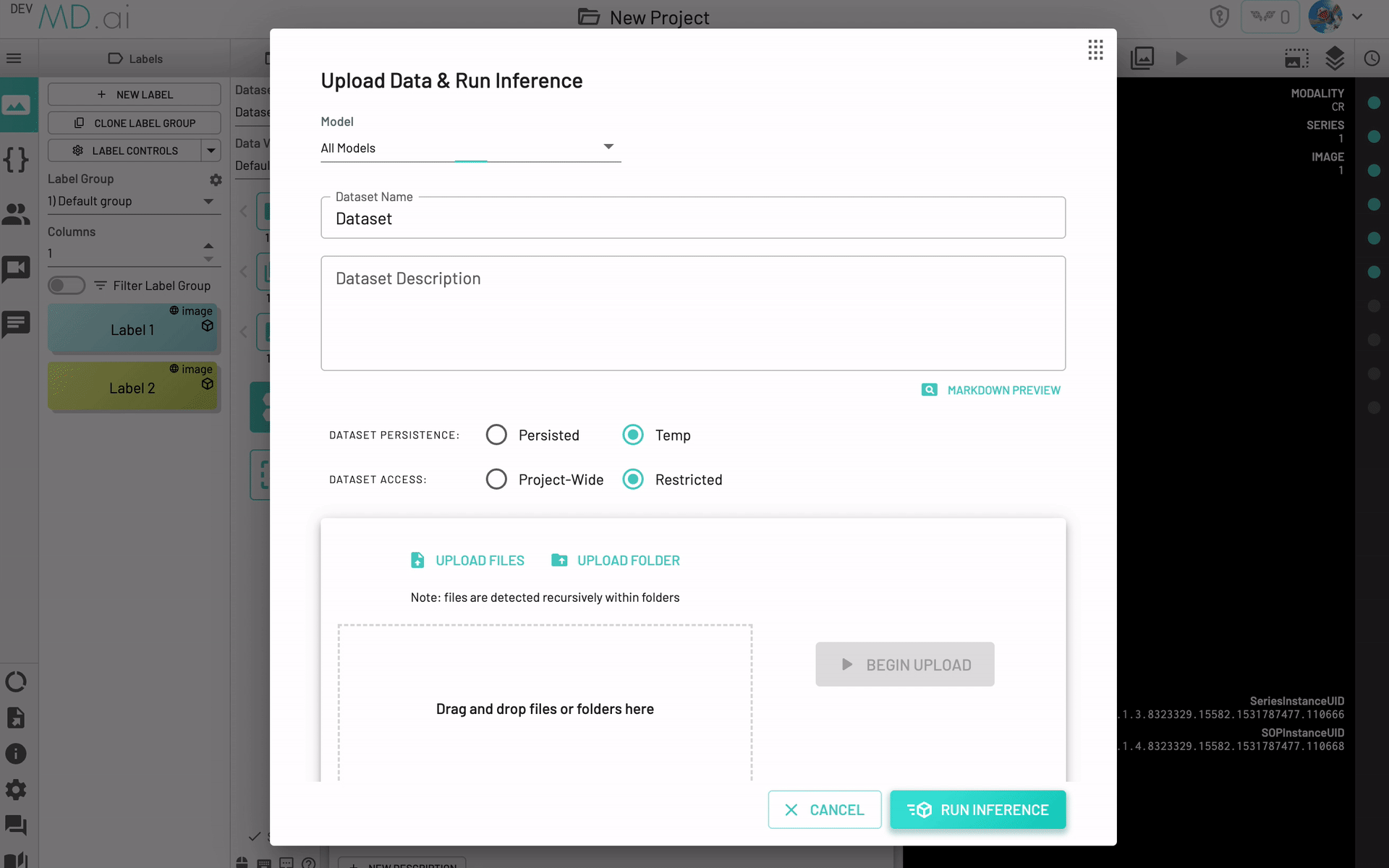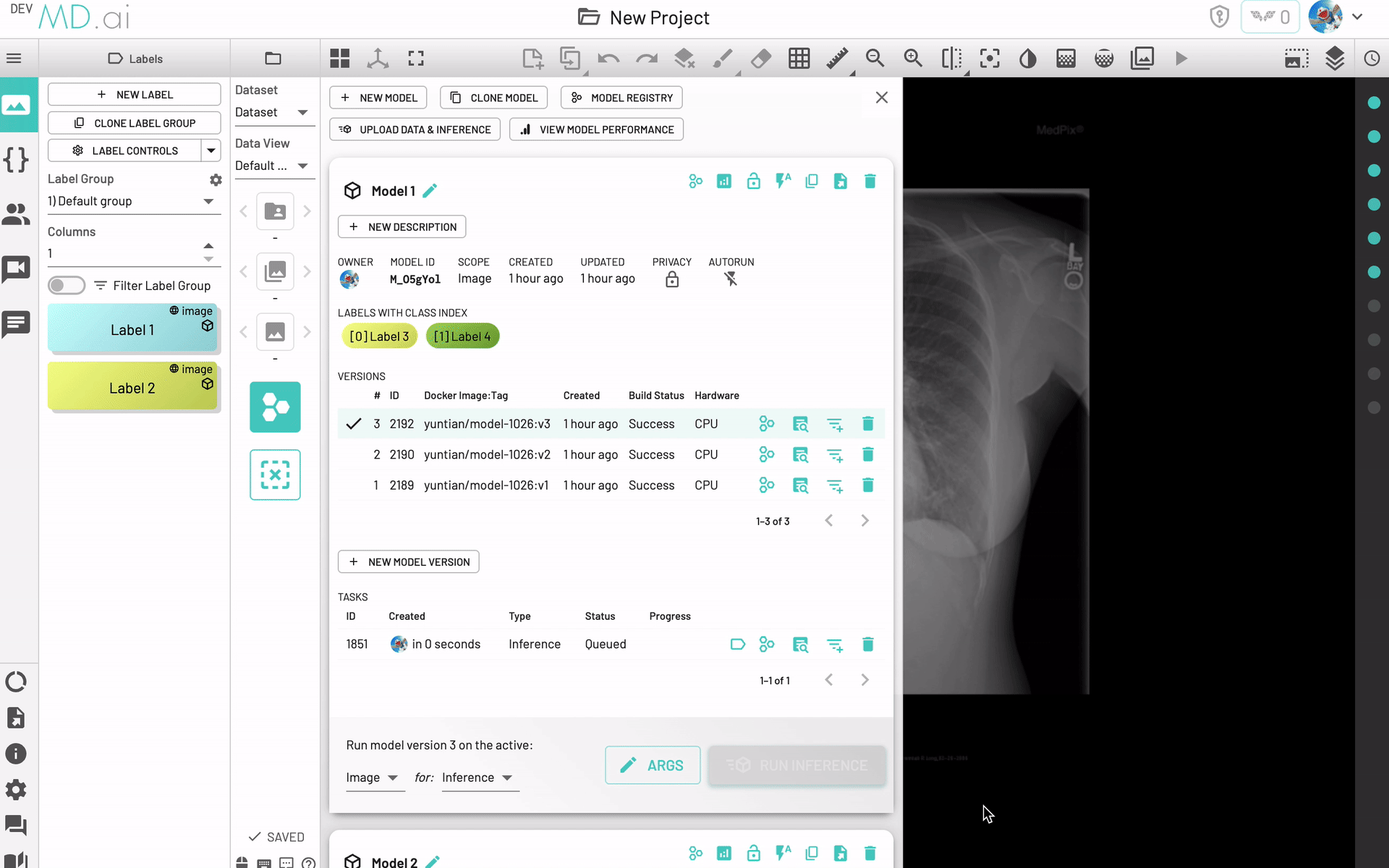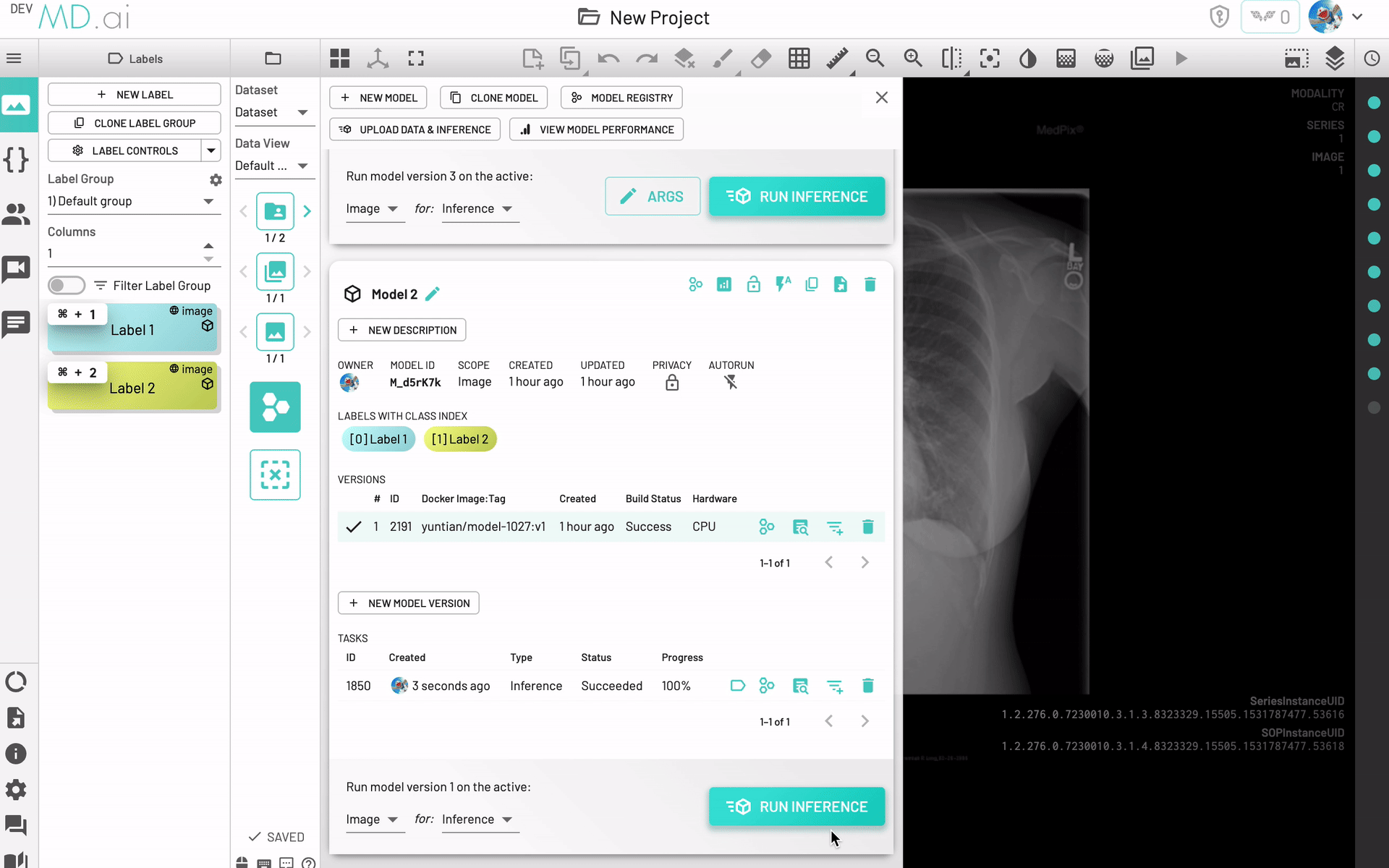
Upload and Inference
Choose this option in the models menu to quickly upload data and run initial inference on the data.
Bug Fixes
- Fixed bug causing empty archive files to prematurely finish upload batch processing.
- Resolved display bug for multi-phasic studies with interleaved slices.
- Fixed navigation panel so multi-frame images now sort by frame number, not instance number from dicom tags.
- Resolved bug preventing model build notification email from reaching user.